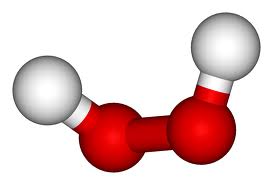
Facts:
2(HO) is the chemical formula for Hydrogen Peroxide
Molar Mass: 34.0147g/mol
melting point: -0.43 degrees Celsius, or 31 degrees Fahrenheit
boiling point: 150.2 degrees Celsius, or 302 degrees Fahrenheit
it is not found in nature, it has to be manufactured
it is considered an oxidant, corrosive, and harmful
Molar Mass: 34.0147g/mol
melting point: -0.43 degrees Celsius, or 31 degrees Fahrenheit
boiling point: 150.2 degrees Celsius, or 302 degrees Fahrenheit
it is not found in nature, it has to be manufactured
it is considered an oxidant, corrosive, and harmful
History/info:
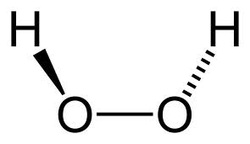
Hydrogen Peroxide is the simplest peroxide (a compound with an oxygen-oxygen single bond) and an oxidizer. hydrogen peroxide is a clear, viscous liquid. Its oxidizing capacity is so strong that it is considered a highly reactive oxygen species. it is considered important because of its oxidizing properties.
Hydrogen peroxide was first described 1818 by Louis Jacques Thenard by reacting barium peroxide with nitric acid. Method of production stayed the same until the middle of the 20th century, where it transitioned into a much more complicated process called the anthraquinone process. it is 2 oxygens sharing a single bind with a hydrogen bonded to each oxygen.
Hydrogen peroxide was first described 1818 by Louis Jacques Thenard by reacting barium peroxide with nitric acid. Method of production stayed the same until the middle of the 20th century, where it transitioned into a much more complicated process called the anthraquinone process. it is 2 oxygens sharing a single bind with a hydrogen bonded to each oxygen.
Uses:
-making of rocket propellant in high concentration as a monopropellant or as the oxidizer component in a bipropellant rocket fuel.
-waste water treatment were it is used as an oxidizing agent on easily oxidized polutants
-Controlling hydrogen cyanide in municipal sludge
-oxygenation
-as a product that prevents infection transmit transmition in the hospital environment
-alternative uses including drinking diluted H2O2 to treat diseases, or as a toothpaste
-waste water treatment were it is used as an oxidizing agent on easily oxidized polutants
-Controlling hydrogen cyanide in municipal sludge
-oxygenation
-as a product that prevents infection transmit transmition in the hospital environment
-alternative uses including drinking diluted H2O2 to treat diseases, or as a toothpaste
Reactions:
Decomposition: Hydrogen Peroxide will spontaneously decompose into water and oxygen gas. 2 h202->2 h20+o2. this is why high concentration hydrogen peroxide should be refrigerated, because refrigeration slows down this reaction.
Links:
This website was created by Tomas Beer